Analysis sent out every Monday
Build with Axial: https://axial22.axialvc.com/
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Cell reprogramming
Cell reprogramming holds the promise of taking any cell and transforming it into a neuron to treat Parkinson’s or a β-islet to cure T1D. A lot of progress has happened in the last 20 years making cell reprogramming one of the fastest moving fields in life sciences. Significant challenges remain to translate discoveries into safe medicines. The field is highlighted by old technologies for new problems and monopolies for new technologies. Cell reprogramming could unlock the development of stem cell therapies for everything from Parkinson’s to cancer and diabetes. With companies like Cellforma, Cellino, Bit.bio, Mogrify, Stately Bio emerging and Fate Therapeutics as a pioneer, there are countless opportunities to generate high-value cell lines for drug development, research, tools, and more. There are 3 main approaches in the field:
Genetic factors to induce stem cell differentiation
Using CRISPR to activate a set of genes to reprogram cells
Small molecule initiated differentiation
The key moment in the field was the 2006 publication by Kazutoshi Takahashi and Shinya Yamanaka discovering 4 transcription factors (Oct3/4, Sox2, Klf4 and c-Myc), now called Yamanaka factors, that convert somatic cells to pluripotent ones. Like most great discoveries, Yamanaka and his lab had to overcome funding challenges, discouragement from colleagues, and very hard technical challenges. Along his journey, Yamanaka considered quitting science and going back to medicine as a surgeon. A decision particularly hard to resist given his father wanted Shinya to be a doctor. In 1996 after a postdoc at the Gladstone (UCSF) where he studied c-Myc, Shinya and his family returned to his hometown in Japan to become a professor at Osaka City University. Every few years, his lab would move to a new Japanese university. However, his group’s research focus never changed despite various struggles: generating embryonic stem cell-like cells from somatic cells, without an embryo.
Tomoko Ichisaka, a long-time research technician from the Yamanaka Lab, would play a key role during this story. A mouse model (Fbx15 knockout) he generated early-on would play the pivotal part years later to screen for the factors that generate induced pluripotent stem cells (iPSC). Yamanaka’s initial hypothesis was that factors that maintain pluripotency in stem cells might also induce it in somatic cells. This thread led to the discovery of 24 candidate genes by 2004. The Fbx15-knockout mouse became particularly useful around this time: Fbx15 is specifically expressed in mouse embryonic stem cells so Yamanaka’s group knocked a gene for antibiotic resistance (neoR) into the Fbx15 locus, which protects stem cells from the antibiotics but makes somatic cell vulnerable. So if a somatic cell became pluripotent after exposure to one/some of the 24 factors, it would be resistant to the antibiotic. This key experiment found that delivering the entire set of 24 genes with retroviral vectors, but not one-by-one, generated several antibiotic-resistant colonies of fibroblasts. Follow-up experiments culminating in 2005 found 4 transcription factors, Oct3/4, Sox2, Klf4 and c-Myc (also goes by OSKM), were essential. After making sure this data was reproducible, they published the work in Cell in 2006. Showing, for the first time, the ability to generate pluripotent stem cells from somatic ones.
Just 4 factors were needed to reprogram cells to a pluripotent state. The simplicity was surprising but once the work was reproduced in other labs, the method took the stem cell field by storm and became the standard way to generate stem cell lines. In 2007, Yamanaka’s lab reported the generation of human iPSCs from human fibroblasts. And labs around the world built upon this discovery and developed new factor combinations for reprogramming and so much more. In 2012, Yamanaka along with John Gurdon (who first showed nuclear reprogramming with Xenopus), won the Nobel Prize. We can all be thankful that Yamanaka persisted despite a ~decade of struggling and decided to not become a surgeon like his father wanted & stayed the course with his scientific work.
Cells exist in various states with 3 main ones:
Pluripotent = can become a type of cell
Multipotent = can become a type of cell within a particular lineage
Terminal = differentiated into something like a neuron or fibroblast
In 1957, Conrad Waddington described embryonic development as a ball rolling down the slope of a hill through a single path and becoming progressively more restricted as it travels from top-to-bottom. Similarly, cell fates follow a unique path that differentiates them from other cell types. Yamanaka’s work was the culmination of ~2 decades worth of research finding that the ball can be rolled back up the hill and revert a terminal cell to an earlier stage.
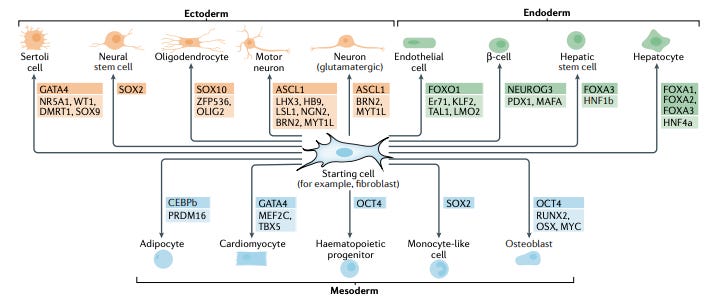
The reprogramming field is centered around genetic factors. Work over the last 15 years or so has led to various formulas to convert cells into other types. The advent of CRISPR in 2012 made gene editing more accessible as a means to reprogram cells. And finally, small molecules have become a more viable approach recently:
Genetic factors = the human genome encodes over 1500 transcription factors (TF) and they can be screened (with retroviruses, mRNA, nanoparticles) to identify which combinations can generate a given cell type. Transcriptomic information across different cell/tissue types is useful to discover which TFs are differentially expressed across various cell states. However, differential expression is often not enough and other methods need to be used. Actually, work in cell reprogramming led to the formation of Moderna - in 2010, mRNA was used to induce pluripotency in fibroblasts providing an efficient and safer way to generate iPSCs and also sparked the formation of the mRNA drug developer.
CRISPR = relies on fusing a dead Cas9 (dCas9) to transcription activators/repressors, epigenetic remodeling factors, among others for CRISPR activation (CRISPRa) screens. The endogenous expression genes of TFs can be activated often with a higher reprogramming efficiency (80% versus say 20% in a lot of cases) than genetic factors. However, pooled CRISPRa screens produce a significant number of false-positives/negatives.
Small molecules = chemicals like retinoic acid, nicotinamide, tranylcypromine, and TGF-β inhibitors have been used to reprogram cells. This approach started gaining traction around 2013 and has delivery advantages and the ability to work in vivo, but has issues with differentiation efficiency. Recent research published earlier in 2022, excitedly reported induced pluripotency of somatic cells purely through chemical reprogramming.
All 3 approaches share the same goal - determine a formula that can convert a starting cell type to another one. Each cell contains the same set of >20K genes so their identity is determined by how these genes are expressed and function. Each of these methods work to modify the epigenetic landscape of a cell and shift gene expression profiles to guide a cell through a particular trajectory ending at a new cell state. This can be done indirectly with an iPSC as the intermediary. However, direct reprogramming, say a fibroblast to a neuron, is preferred due to the protocol’s simplicity:
Indirect reprogramming = a somatic cell is converted into an iPSC, which is then guided to become a new cell type. This approach involves an intermediate pluripotent state that is more straightforward to execute but requires ex vivo cell expansion/transplantation.
Direct = allows for the conversion of a cell type in situ without a transition state, but efficiency rates are relatively lower and there are delivery risks
On a side note, partial reprogramming has also been found to reduce signs of cellular aging without a loss in identity. Juan Carlos Izpisua Belmonte’s lab, formerly at the Salk & now he’s at Altos Labs, has been the pioneer publishing a key paper in 2016 that showed transient, periodic induction of OSKM (2 days on, 5 days off and repeated several times) can alleviate age-associated hallmarks.
On the commercial-side, many companies have tried to develop new stem cell therapeutics with little success so far. The only exception are bone marrow transplants. But they have been performed since the 1960s to cure various blood diseases but are more similar to a full organ transplant than a cell therapy. For a BMT, the entire hematopoietic system is replaced after irradiation of the patient’s old hematopoietic system. The comparable couldn’t be done for a neuron or lung cell transplant. Barring complete removal of previous cells, drug development with cell reprogramming will need to figure out ways to ensure engraftment & purity and reduce the risks of teratoma formation. First you have to figure out a way to make sure the cells designed don’t initiate graft versus host disease (GvHD). Autologous (from the same patient) offers advantages here but increases manufacturing costs and turnaround time. The other alternative, allogeneic (off-the-shelf) solves those problems but may always be relegated as a bridging therapy rather than a curative one due to long-term safety risks. After a set of cells are differentiated, you have to make sure there aren’t any pluripotent ones left over - they can lead to tumor formation and also other things like bone cells growing in your eye. Purity in stem cell therapeutics becomes the most important problem to solve. And this is why direct reprogramming is preferred. The indirect approach will require new tools to filter out pluripotent cells before transplantation into a human. While more work needs to be done to increase the efficiency rates of direct reprogramming.
Fate Therapeutics, the most successful company in the field, was founded in 2007 to take recent work around cell reprogramming and develop drugs. With foundational IP from Rudolf Jaenisch’s lab, which had the earliest art that describes reprogramming a human somatic cell to pluripotency, the company had to make several pivots due to the difficulties of developing stem cell therapeutics. Their first product was a blood transplant, ProHema but after a few iterations, they are currently focusing on NK cells. Fate’s programs have always been exciting and their recent success in cancer helps validate iPSCs as a platform. Recent data from Fate opens up the questions of how to maximize efficacy of these cells. This makes sense given the focus is on developing protocols and editing cells to make sure they are safe while probably reducing their potency. However, Fate did establish that cell reprogramming can play a role in drug development.
There is also Semma Therapeutics developing cell transplants for T1D. The company’s name is a combination of two of Doug Melton’s children’s names: Sam & Emma. Melton has a great story himself - advised by John Gurdon actually during grad school, Melton completely shifted the focus of his lab to type I diabetes decades ago once he learned his children had the disease. It’s taken him almost 3 decades, but he’s at the door of nearly curing it. He co-founded Semma, which was then acquired by Vertex who is running a phase 1/2 trial to explore the effects of their reprogrammed β-cells (insulin producer) in T1D patients. Early data (n-of-1) has been exciting and shows a path to curing the disease.
Ultimately, whether it’s full or partial reprogramming, the opportunity is to develop differentiation protocols (indirect & direct) to create any cell type. Making it easy to go from an adipocyte to a neural stem cell. Then that neural stem cell can become a neuron, astrocyte, or an oligodendrocyte. And back.